You take a refreshing sip of a cold soda, a energizing gulp of sports drink, or a soothing swallow of hot lemon water. These beverages are a daily part of life for millions, offering comfort, hydration, or a needed caffeine boost. But what if every sip was launching a silent, chemical attack on the hardest substance in your body? This isn’t a story about sugar and cavities; it’s a more insidious process called dental erosion—the direct, irreversible dissolution of your tooth enamel caused by acid. Unlike decay, which is a bacterial process, erosion is a simple chemical one: acid wins, enamel loses. This article will serve as a stark wake-up call, revealing the hidden acidity in common drinks, explaining the precise science of how they destroy enamel, and providing a practical battle plan to protect your smile from this invisible threat.
1. The Chemistry of Destruction: Understanding pH and Enamel
To grasp erosion, you must first understand pH. The pH scale measures how acidic or basic a substance is, running from 0 (extremely acidic) to 14 (extremely basic), with 7 being neutral.
The Critical Threshold: pH 5.5
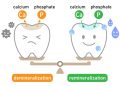
Tooth enamel, while the hardest substance in the human body, is primarily composed of a mineral called hydroxyapatite. This mineral is highly susceptible to acid. The “critical pH” at which enamel begins to demineralize and dissolve is approximately 5.5. Any beverage with a pH lower than 5.5 has the direct chemical potential to erode your teeth. The lower the pH, the more aggressive the attack.
How Erosion Differs from Decay:
- Dental Erosion: A chemical process. Acid directly dissolves the enamel surface. It smooths out the anatomy of the tooth, making it look glossy, creates cupping on the chewing surfaces, and thins the edges, making them appear translucent and sharp.
- Dental Caries (Cavities): A bacterial process. Bacteria in plaque consume sugar and produce acid as a waste product. This acid creates a localized hole or pit in the tooth.
Erosion often paves the way for decay by wearing away the protective enamel shell, making the underlying, softer dentin more vulnerable.
2. The Usual Suspects: A List of the Worst Acidic Offenders
While sour candies and vinegar are obvious acids, many popular drinks are deceptively corrosive. Here are the worst offenders, often with pH levels far more dangerous than you might assume.
The Top Tier (Extremely Acidic – pH below 3.0):
- Soft Drinks (Regular and Diet): This is the arch-nemesis of enamel. The phosphoric acid in colas (pH around 2.4-2.5) and the citric acid in citrus-flavored sodas (like Sprite or Mountain Dew, pH around 3.0-3.5) are exceptionally corrosive. Crucially, diet sodas are just as erosive as regular sodas. The acid is the problem, not the sugar.
- Energy Drinks: Marketed for performance, these are often among the most acidic beverages available, with some brands having a pH as low as 2.0-2.5. They combine citric and phosphoric acid with high sugar content, delivering a double blow of erosion and decay.
- Pure Lemon Juice (pH ~2.0): Often promoted in “detox” or “wellness” shots, straight lemon juice is pure citric acid and is devastating to enamel.
The Middle Tier (Highly Acidic – pH 3.0 to 4.0):
- Fruit Juices: Apple juice (pH ~3.4), orange juice (pH ~3.7), and grapefruit juice are all highly acidic due to their natural fruit acid content.
- Sports Drinks: Designed to rehydrate, their citric acid content (pH ~3.0-3.5) bathes the teeth in acid, especially during physical activity when saliva flow may be reduced.
- Flavored Waters and Iced Teas: Many commercially flavored waters and bottled iced teas contain citric or other acids as preservatives and flavor enhancers, pushing their pH into the danger zone.
- White Wine (pH ~3.0-3.5): Generally more acidic than red wine, making it a significant erosive risk.
The Surprise Culprits (Moderately Acidic – pH 4.0 to 5.5):
- Black Coffee (pH ~4.7-5.0): While not as aggressive as sodas, its acidic nature, combined with frequent, all-day sipping, can contribute to significant enamel wear over time.
- Beer (pH ~4.0-4.5): Certain styles, especially sour beers, can be quite acidic.
3. The Slow Fade: The Timeline and Progression of Erosion
Dental erosion is not a single-event catastrophe; it’s a slow, cumulative process of destruction that occurs over months and years.
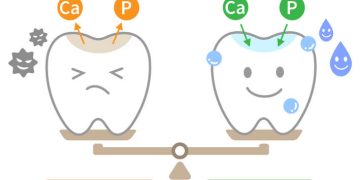
The Immediate Attack (Within Seconds): The moment an acidic drink enters your mouth, the pH on the tooth surface plummets. The hydroxyapatite crystals in the enamel begin to dissolve immediately. The surface becomes microscopically softer.
The Salivary Defense (20-30 Minutes): This is your mouth’s natural recovery system. Saliva is slightly basic (pH ~7.4) and works to neutralize acids, wash them away, and provide calcium and phosphate to aid in “remineralization”—the natural repair of early erosion. However, this process takes time. If you sip an acidic drink slowly over an hour, you are re-acidifying your mouth every few minutes, never giving saliva a chance to do its job.
The Long-Term Consequences (Months to Years):
- Early Stage: The enamel surface loses its microscopic texture, becoming smooth and glossy. The edges of the front teeth may start to appear slightly translucent.
- Moderate Stage: The enamel is visibly thinned. The chewing surfaces of the back teeth develop small, shiny dents (cupping). Discoloration occurs as the yellower dentin layer becomes more visible through the thinning enamel.
- Severe Stage: Enamel is lost entirely on some surfaces, exposing the sensitive dentin. Teeth become shorter, appear worn down, and are highly sensitive to temperature and touch. The biting edges of the front teeth may chip or crack easily because they are so thin.

4. Fortifying Your Defenses: Proactive Prevention Strategies
The best way to deal with erosion is to prevent it from happening. A few simple changes in habit can make a world of difference.
Rethink Your Drinking Habits:
- Limit Frequency: This is the most critical rule. It is far better to drink one can of soda in 5 minutes than to sip on it for 3 hours. Constant sipping creates a perpetual acid bath.
- Use a Straw: Position the straw toward the back of your mouth to bypass your front teeth, minimizing direct contact with the liquid.
- Drink Quickly: Consume acidic beverages in a short period rather than nursing them.
Timing is Everything:
- Drink with Meals: Consuming acidic drinks with food helps neutralize the acid more quickly because chewing stimulates protective saliva flow.
- Avoid Brushing Immediately After: This is a classic mistake. The acid softens the enamel, and brushing right away can actually brush the enamel away. Wait at least 30-60 minutes after an acidic drink or meal to allow saliva to re-harden the enamel surface.
Strengthen Your Enamel:
- Use Fluoride: Fluoride is a superhero for your enamel. It incorporates into the tooth structure to form fluorapatite, a crystal that is more resistant to acid than natural hydroxyapatite. Use a fluoride toothpaste and consider an over-the-counter fluoride rinse.
- Choose Water: Make water your default, go-to beverage. It has a neutral pH, helps rinse away acids and sugars, and contains no calories.
5. Damage Control: Effective Neutralizing and Remineralizing Strategies
When you do consume something acidic, you can take active steps to mitigate the damage and help your teeth recover.
The Immediate Rinse: Swish your mouth with plain water immediately after finishing the drink. This helps to physically wash away the acid and raise the oral pH rapidly.
The Cheese Strategy: If available, eat a piece of cheese after a meal or drink. Cheese is basic and helps neutralize acid. It also stimulates saliva flow and provides a dose of calcium and phosphate for remineralization.
Sugar-Free Gum: Chewing sugar-free gum, especially one containing xylitol, is an excellent post-meal habit. It boosts saliva flow by a factor of ten, accelerating the neutralization process and bathing the teeth in reparative minerals.
Professional Interventions: If you are at high risk for erosion, talk to your dentist. They can provide:
- High-Fluoride Prescription Toothpaste: These have a higher concentration of fluoride to aggressively strengthen enamel.
- Remineralizing Agents: Products like CPP-ACP (Recaldent) found in some gums and toothpastes can provide a super-saturated dose of calcium and phosphate to the tooth surface, enhancing repair.
The threat posed by acidic drinks is real, silent, and cumulative. But it is not inevitable. By moving from unconscious consumption to informed action, you can take control of your oral environment. Understand the pH of your favorite drinks, change how you consume them, and actively support your mouth’s natural defense systems. Your smile is worth protecting. Choose your drinks wisely, and never underestimate the power of a simple glass of water.

























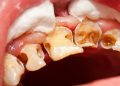
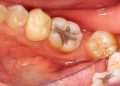



Discussion about this post